KANSAS CITY, MO—Disruptive clumps of mutated protein are often blamed for clogging cells and interfering with brain function in patients with the neurodegenerative diseases known as spinocerebellar ataxias. But a new study in fruit flies suggests that for at least one of these diseases, the defective proteins may not need to form clumps to do harm.
The study, published February 1, 2014, in the journal Genes and Development, focuses on ataxin-7, the gene that is mutated in patients with spinocerebellar ataxia type 7 (SCA-7). Researchers led by investigators Jerry Workman, Ph.D. and Susan Abmayr, Ph.D. at the Stowers Institute for Medical Research found that fruit flies that lack Ataxin-7 experience neurodegeneration in the brain and the eye—paralleling the effects of the human disease. "The assumption has been that the disease is caused by the aggregated proteins," Workman says. "But in the mutated fly, there's no aggregated protein. There's no soluble protein. It's not there at all. The lack of Ataxin-7 causes neurodegeneration in the fruit fly."
Workman and Abmayr did not set out to study Ataxin-7. For more than a decade, the husband-and-wife team has been investigating how a large protein complex called SAGA, which helps control gene activity in organisms from yeast to humans, influences developmental processes. Workman's lab discovered the complex in yeast in the 1990s. In fruit flies, about 20 different proteins come together to form SAGA, which modifies DNA packaging in multiple ways to influence the activity of thousands of genes.
When Vikki Weake, Ph.D., a former postdoctoral researcher in Workman's lab, began investigating the components of the fruit fly SAGA complex, she came across a protein that had not previously been studied in fruit flies. Postdoctoral researcher Ryan Mohan, Ph.D., focused his attention on that novel protein, and by scouring genetic databases, uncovered a resemblance to human Ataxin-7.
Ataxin-7 is one of several genes associated with neurodegenerative diseases, including Huntington's disease and other spinocerebellar ataxias. These genes sometimes develop a genetic "stutter" in which a three-letter segment of the DNA code is repeated over and over. "It's not known how that affects that activity of the protein," Abmayr says. "But what is known is that it can cause aggregation." The expansion in the genetic code leads to proteins containing long, redundant strings of a single amino acid called glutamine, she explains. These abnormal proteins are prone to aggregating with one another inside cells.
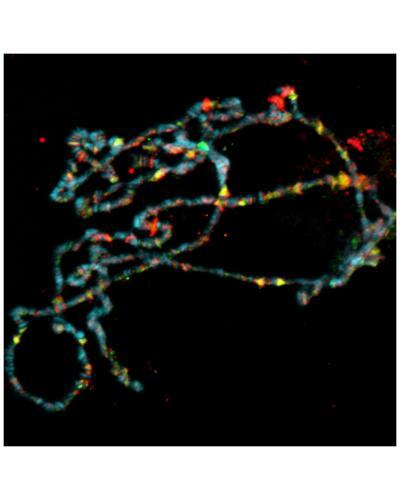
Ataxin-7, one of several genes associated with neurodegenerative disease, anchors a key enzymatic module to the fruit fly SAGA complex, which helps control gene activity in organisms from yeast to humans. Fly polytene chromosome preparation with Ataxin-7 is shown in green, RNA polymerase in red and DNA in blue.
(Photo Credit: Image courtesy of Dr. Ryan Mohan of Stowers Institute for Medical Research)
Ataxin-7 was known to aggregate in the cells of patients with SCA-7, but there was no direct evidence linking the aggregates to neurodegeneration. So when the team stumbled across a version of Ataxin-7 in fruit flies, they saw an opportunity to learn more.
Mohan says most studies of ataxins and related proteins had focused on the effects of the polyglutamine-expanded versions, rather than the function of the unaltered protein. "I considered that this was part of a larger protein complex that was regulating many, many genes, and I thought it was important to find out what the protein normally does to regulate the complex," he says. "From that position, I could consider what a polyglutamine insertion might do to disturb those functions."
Mohan conducted detailed biochemical analyses to better understand how loss of Ataxin-7 affected the SAGA complex. His experiments showed that Ataxin-7 anchors one of the complex's enzymatic modules, which is responsible for removing chemical tags called ubiquitin from DNA-packing proteins. Without Ataxin-7, this module falls off the complex. On its own, the released module becomes overactive, removing too many ubiquitin tags. This can lead to misregulation of genes. The scientists are planning further experiments to find out which genes are affected when Ataxin-7 stops working.
Next, Mohan genetically engineered flies without any Ataxin-7. Without the protein, most flies died as embryos. Those that survived to adulthood had movement problems, demonstrated by their inability to climb. When the scientists looked at the structure of the neurons in the insects' brains and eyes, they saw that while the tissue in very young flies was more or less intact, problems developed quickly. "They are initially kind of okay, but after a few days we see massive degeneration in the brain and the eye," Abmayr explains. Similar effects were seen in flies whose Ataxin-7 gene was shut off only in cells in the brain and eyes. The neurodegeneration that the scientists observed was similar to what other researchers had found in flies producing a human Ataxin-7 protein with the polyglutamine expansion.
"This sheds new light on what we think could be the cause of the SCA7 disease phenotype," Abmayr says. "The problems associated with the disease may be because these polyglutamines actually knock out the function of the Ataxin-7 protein."
The fruit fly has the potential to reveal more about Ataxin-7's role in disease, say the scientists, who are now planning to create a mutant fly whose Ataxin-7 contains a polyglutamine expansion. "Hopefully with a combination of biochemistry and genetics we can sort out how much of the neurodegeneration comes from the fact that you don't have the normal Ataxin-7 protein, and how much is because of the aggregated protein," Abmayr says.